nextaro® Transfer Devices – one family with hundreds of products
A wide spectrum of vial and stopper sizes, geometries and material properties define the real‑world complexity of pharmaceutical transfer applications. Built on a modular platform approach, nextaro® enables structured variant creation to address this variability. Variants can be configured across product type, venting concept, swabable valve options, vial sizes, in-line filtration levels and market‑specific labeling. In addition, performance characteristics such as filter retention size, transfer speed or residual volume can be tailored based on requirements derived from the pharmaceutical formulation itself and the way the product is handled by the end user.

nextaro® Transfer Devices – FDA‑ready by design
Customized transfer devices are often associated with extended development timelines and regulatory uncertainty. Sfm has proven that this doesn’t have to be the case. The nextaro® product family was intentionally designed to address this challenge through a platform-based development and documentation strategy aligned with FDA Design Control expectations.
Platform consistency enables speed
All nextaro® variants are based on identical materials in the fluid path, a shared functional architecture and a standardized use case envelope as transfer devices according to ISO 22413. As a result, existing biocompatibility according to ISO 10993 and material safety data can be transferred to new variants. This substantially reduces development effort, testing scope (time and costs) and regulatory complexity
Standardized, Norm‑Based Testing Strategy
All performance, safety and functional testing is conducted using internally defined, standardized test procedures that are aligned with applicable international standards (incl. ISO 8536-4, USP 788, ISO 11607 and more), consistently applied across the product family, and validated through repeated regulatory usage. New variants therefore follow established test pathways, not ad hoc or project specific test concepts.

FDA‑Compatible Design Control & Documentation Structure
The nextaro® Design History File (DHF) is structured in accordance with ISO 13485 design control principles, and FDA design control expectations (21 CFR 820). Key characteristics are modular documentation blocks, traceable requirements, verification and risk links and change oriented variant extensions instead of new DHFs. The risk management system according to ISO 14971 is deliberately structured to allow low effort extension to additional variants, rather than re initiation of risk analyses.
Proven Regulatory Practice – Not Theory
This platform-based approach has been successfully applied multiple times across the nextaro® family. From initial FDA compliant re documentation, through multiple product and size variants to market access in regulated regions, including the U.S.. The regulatory strategy is therefore based on execution experience, not assumptions.
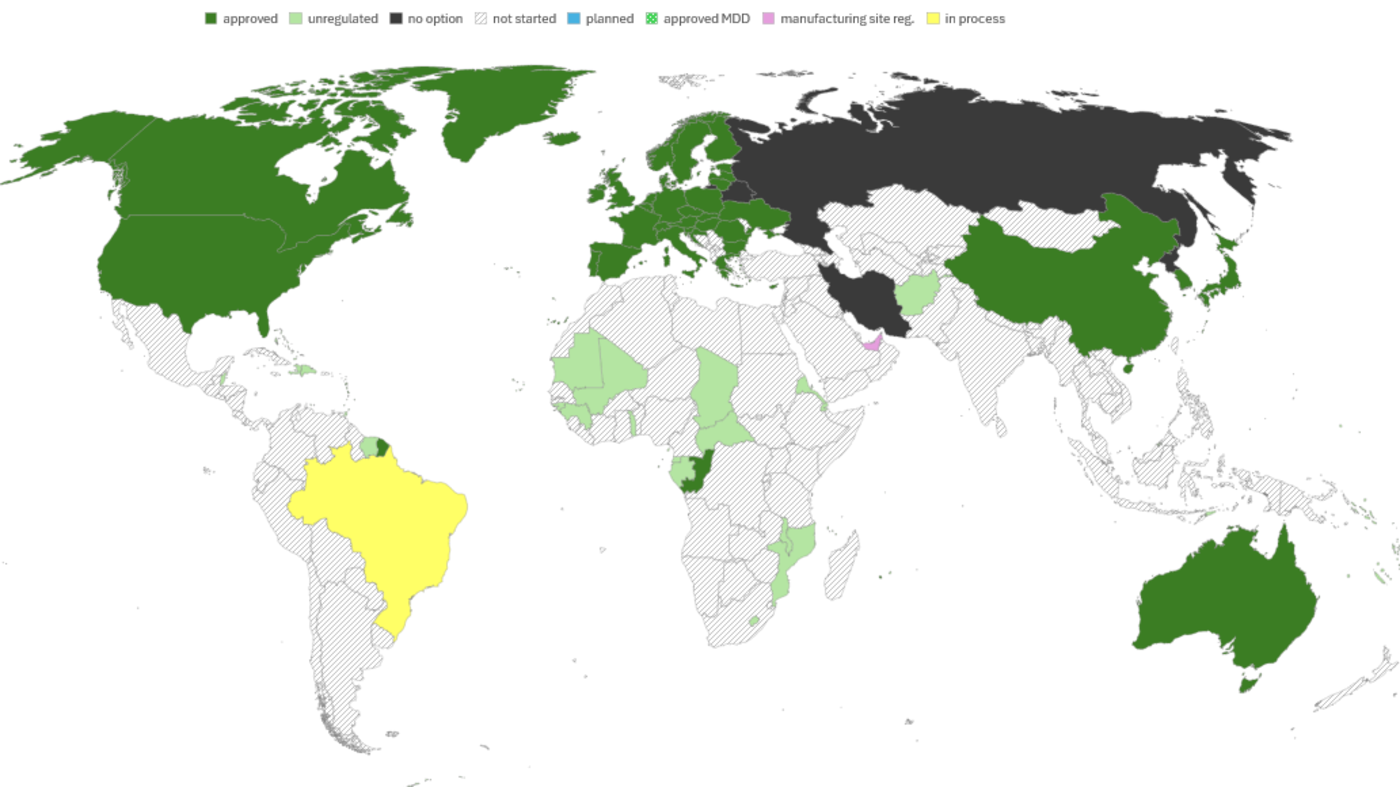
510(k) Strategy: Variant Assignment Instead of New Submissions
Due to the broad functional and technical span of the existing nextaro® family, new variants can often be assigned to an existing 510(k) as a configuration or size/material variation rather than triggering a new submission. This approach is supported by identical intended use, unchanged fundamental technology, comparable materials and fluid path, and well-defined design controls. Of course, this is subject to an individual regulatory assessment per variant and customer use case.
Which parameters matter most for your transfer device?
Get in touch with us to explore how the nextaro® platform can be configured to your needs.