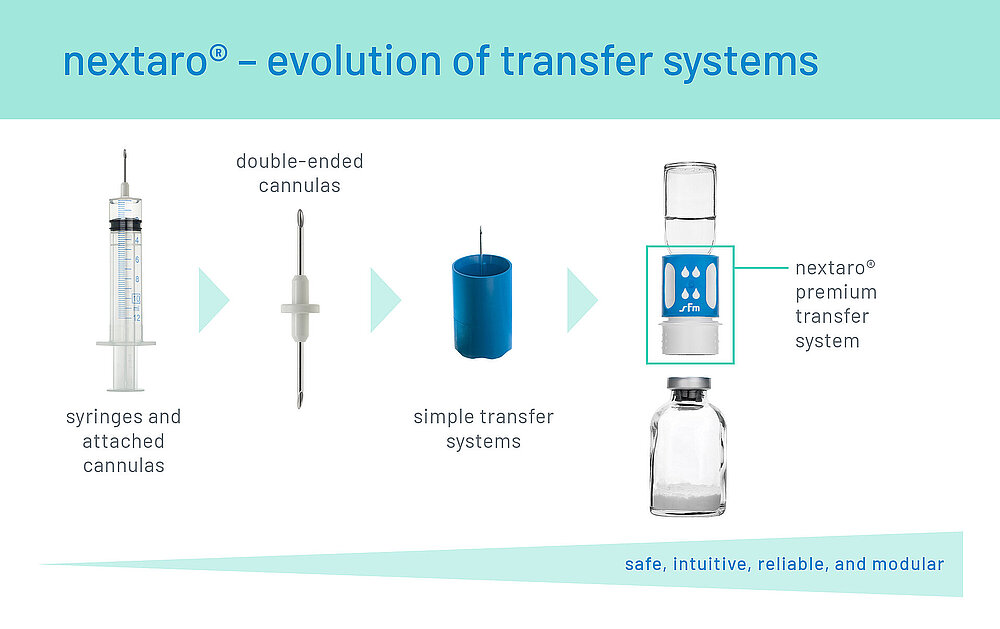
Evolution of transfer systems

Simple systems for reconstitution
Reconstitution and absorption of lyophilized active ingredients was previously carried out with syringes and attached cannulas. The septum of the vial was pierced directly with the cannula. However, there was a high risk of injury to medical staff due to the sharp, unprotected metal cannulas and impractical handling. The rubber stopper could also be sheared off and the active ingredient contaminated, depending on the geometry and design of the cannula.
Development of complex transfer systems
This led to the development of double-ended cannulas and simple transfer systems with double spikes made of plastic. In addition, filters were increasingly used to protect the active ingredient from contamination, initially as separate syringe filters and later as integrated components of the transfer systems.
Modern transfer systems for reconstitution ensure safe, intuitive and easy handling for the user, and they are continuously developed and optimized in order to meet the requirements of high-quality and valuable active ingredients (Fig. 1).
Requirements for transfer systems
Patient safety, user protection and high pharmaceutical quality are key issues for reconstitution and transfer systems. This requires that:
- The transfer system is seated securely and stably on the vial
- The lyophilized active ingredient can be reconstituted according to the particular requirements
- Particle contamination of the medication is avoided, for example when the septum is pierced
- The remaining volume is minimized and the quantity of medication is sufficient
nextaro® - the premium transfer system sets the standards
The product line based on the nextaro® transfer system from sfm medical devices goes one step further and offers additional advantages:
- Intuitive handling: with its intelligent packaging, well thought-out design and optimized feel, nextaro® enables simple and reliable use in the clinic and home care sectors.
- High safety level: theplastic spikes are located in the housing, providing the user with optimal prevention against injuries.
- High quality and availability of the reconstituted active ingredients: the use of two filters in the nextaro® transfer system protects against contamination at every stage. The patented rotation device ensures an absolutely tight system.
- Robust and stable construction: the specially designed product configurations and materials are of the highest quality and give the user a feeling of absolute security and control.
- Modular system: nextaro® can be adapted to the individual requirements of various active ingredients and solvents.
The impressive development of the nextaro® premium transfer system shows that the evolution of reconstitution systems is well advanced. Well thought-out innovations in the nextaro® premium transfer system product line enable individual adaptations to current as well as new requirements. The nextaro® product family will continue to grow in the future in order to meet the requirements of the market with its revolutionary solutions — our focus is always on patients and users.